55503281
Kohlenhydrate: zuckersüße Chemie
In 10 interaktiven H5P-Modulen wird Wissen zum Thema Kohlenhydrate vermittelt und anschließend abgefragt.
Das Medium bietet H5P-Aufgaben an, die ohne zusätzliche Software verwendbar sind.
Durch interaktive Aufgabentypen wird das audiovisuelle und interaktive Lernen einfach.
Lernen macht jetzt Spaß!
Included Tasks
- I Nahrungsgrundlage: Kohlenhydrate - Aufgaben mit Video
- II Energiekreislauf - interaktive Aufgabe
- III Zucker ist nicht gleich ZUCKER - interaktive Aufgabe
- IV Aufbau der Kohlenhydrate - Lückentext
- V Fehling-Probe - interaktives Video
- VI Einteilung der Kohlenhydrate - interaktive Aufgabe
- VII Einfachzucker: Monosaccharide - interaktive Aufgabe
- VIII Zweifachzucker: Disaccharide - interaktive Aufgabe
- IX Stärkenachweis - Video mit Aufgaben
- X Kohlenhydrate-Quiz - interaktive Aufgabe
Curriculum-centred and oriented towards educational standards
Matching
Acids and Bases
We can find acids and bases in every supermarket, some of them in our food, others in cleaning agents. In everyday products, acids and bases as well as acidic and alkaline reacting salts have extremely different functions. In food, acids are either present or added as flavouring agents such as citric acid, tartaric acid and acetic acid, as antioxidants such as ascorbic acid or generally as acidifiers, sequestrants (citric acid and tartaric acid) and preservatives (acetic acid).
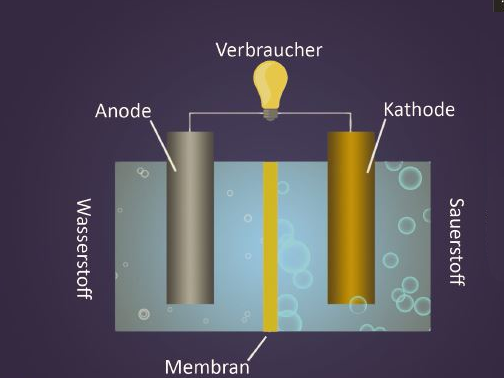
Fuel Cell
A smartphone offers a lot of opportunities nowadays. The numerous apps and applications may enrich your daily life but cost a lot of electricity. It is particularly annoying when the device fails at the most inconvenient moments. Conventional rechargeable batteries are often empty after one day already, and the device needs to be plugged in. Besides many others, also this problem could be solved by using fuel cells – thus considerably increasing the duration of the smartphone.
Basics of Chemistry I
We are surrounded by objects and substances. We recognise objects that are to serve a specific purpose by their shapes. Similar objects may consist of different materials or substances. Substances, however, are independent of shapes and possess very specific properties. We are able to perceive many of these substances with our senses. For example, we can see, touch or smell them so as to be able to recognise them. Chemists are particularly interested in those substance characteristics that can be measured. On the basis of these measurable properties they can distinguish between substances, identify a specific substance or test it for special use. Models help us to understand phenomena. They depict only specific elements of our reality, thus presenting the world in a simplified way. The spherical particle model, for example, helps us to understand how a scent spreads all over the room or substances disperse in water.